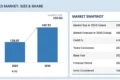
The global In Vitro Diagnostics (IVD) Quality Control Market is projected to reach USD 2.15 billion by 2030, growing from USD 1.65 billion in 2025, at a CAGR of 5.5%. This steady growth is driven by the increasing need for accurate, standardized, and reproducible diagnostic results across clinical settings.
Market Overview
IVD quality control solutions ensure the reliability and accuracy of diagnostic test results. These include quality control products, data management systems, and quality assurance services used across diagnostic platforms such as clinical chemistry, immunoassays, and molecular diagnostics.
The growing number of accredited laboratories and stricter regulatory standards are pushing healthcare providers to adopt robust quality control systems.
Download PDF Brochure: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=198032582
Key Market Drivers
1. Increasing Number of Accredited Clinical Laboratories
Accreditation requirements demand strict adherence to quality standards, boosting demand for reliable QC products and services.
2. Rising Adoption of Third-Party Quality Controls
Third-party controls offer:
- Independent performance validation
- Multi-platform compatibility
- Greater flexibility compared to OEM controls
3. Growing Demand for Diagnostic Testing
An aging population and rising prevalence of chronic and infectious diseases are significantly increasing testing volumes worldwide.
4. Expansion of Point-of-Care (PoC) Testing
The rapid adoption of PoC diagnostics is creating demand for real-time, accurate, and portable quality control solutions.
Market Restraints
Budget Constraints in Healthcare Facilities
Hospitals and laboratories face cost pressures, limiting investment in advanced quality control systems.
Unfavorable Reimbursement Scenarios
Limited reimbursement for diagnostic tests can reduce spending on quality control measures.
Market Opportunities
Rising Demand for Multianalyte Controls
Multi-analyte QC products allow simultaneous testing across parameters, improving efficiency and reducing costs.
Growth in Emerging Markets
Countries in Asia Pacific and other developing regions present strong growth potential due to expanding healthcare infrastructure and diagnostics adoption.
Market Challenges
Stringent Product Approval Processes
Regulatory compliance requirements can delay product launches and increase development costs.
Lack of Standardized Regulations
Inconsistent accreditation standards across emerging economies hinder uniform adoption of QC solutions.
Key Market Insights
- Quality control products dominate the market, including serum/plasma, whole blood, and urine controls.
- Immunoassay segment leads due to high sensitivity, specificity, and reproducibility.
- Third-party controls are the fastest-growing segment due to flexibility and unbiased validation.
- Hospitals are the largest end users due to high testing volumes and regulatory requirements.
- Asia Pacific is the fastest-growing region, driven by rising disease burden and healthcare investments.
Competitive Landscape
Leading players are focusing on strategic collaborations, product innovation, and acquisitions to strengthen their market presence. Key companies include:
- Thermo Fisher Scientific
- Bio-Rad Laboratories
These companies are expanding their portfolios and leveraging partnerships to meet the growing demand for high-quality diagnostic solutions.
Trends & Disruptions in the IVD QC Market
The market is undergoing a transformation driven by evolving laboratory and diagnostic needs:
- Increasing adoption of third-party and multianalyte controls
- Integration of data management systems for real-time QC monitoring
- Growth of point-of-care and rapid diagnostics
- Rising focus on automation and digitalization in laboratories
- Demand for cost-effective and scalable QC solutions
These trends are enabling laboratories and hospitals to improve efficiency, maintain compliance, and deliver faster, more accurate results.
Future Outlook
The IVD quality control market is expected to witness sustained growth due to increasing diagnostic demand, expanding healthcare infrastructure, and technological advancements. The shift toward flexible, independent QC solutions and digital integration will play a crucial role in shaping the market’s future.
Request Sample Pages : https://www.marketsandmarkets.com/requestsampleNew.asp?id=198032582