The United Kingdom cancer biomarkers market is a highly advanced and rapidly expanding sector, driven by the country’s pioneering role in integrating genomics into routine healthcare and its strong emphasis on personalized medicine. The landscape is defined by robust academic-industry collaborations and significant public funding, exemplified by the National Health Service (NHS) Genomic Medicine Service, which has centralized testing into regional Genomic Laboratory Hubs to ensure equitable access to molecular diagnostics. Key growth drivers include the rising incidence of various cancers, particularly breast and lung cancer, and the increasing clinical adoption of sophisticated technologies such as next-generation sequencing and liquid biopsies. While the market faces challenges like high testing costs and complex regulatory requirements, it is bolstered by supportive government initiatives and the integration of artificial intelligence to streamline biomarker discovery and validation. With the market projected to reach approximately USD 2.19 billion by 2026, the UK remains a central hub for oncology innovation, focusing on early disease detection and the development of companion diagnostics to optimize therapeutic outcomes.
Key Drivers, Restraints, Opportunities, and Challenges in the United Kingdom Cancer Biomarkers Market
The United Kingdom cancer biomarkers market is primarily driven by the rising incidence of cancer, a robust academic research base, and significant government support through the NHS, which integrates biomarker testing into standard care pathways for lung, ovarian, and colorectal cancers. Significant growth opportunities exist in the expansion of personalized medicine, the adoption of liquid biopsy for non-invasive monitoring, and the integration of artificial intelligence to improve diagnostic accuracy and clinical utility. However, the market faces restraints such as the high cost of advanced genomic assays, stringent regulatory requirements including compliance with EU Medical Device Regulations, and challenges with reimbursement within the healthcare system. Additionally, the industry must navigate operational challenges, including an acute shortage of skilled pathologists and molecular oncologists, long turnaround times for test results, and the need for standardized clinical validation across fragmented laboratory networks.
Customer Segmentation, Needs, Preferences, and Buying Behavior in the United Kingdom Cancer Biomarkers Market
The target customers for the United Kingdom cancer biomarkers market primarily include hospitals and specialty cancer centers, diagnostic and research laboratories, and pharmaceutical and biotechnology companies. Hospitals and cancer centers represent the largest customer segment, prioritizing reliable, biomarker-based assays that enhance diagnosis, prognosis, and treatment monitoring within clinical workflows to improve patient outcomes. These institutional buyers value integrated diagnostic solutions and precision medicine tools, such as liquid biopsies and next-generation sequencing, which help them navigate complex treatment pathways and meet NHS guidelines for early cancer detection. Purchasing behavior is heavily influenced by government initiatives and public funding, with healthcare providers increasingly preferring high-accuracy multiplex panels and AI-integrated systems that streamline the transition toward outcome-driven care models. Meanwhile, pharmaceutical and biotechnology firms focus on robust biomarker platforms to support companion diagnostics and drug development, often seeking long-term strategic partnerships and large-volume supply contracts with diagnostic providers to accelerate clinical trials and patient stratification.
Regulatory, Technological, and Economic Factors Impacting the United Kingdom Cancer Biomarkers Market
The United Kingdom cancer biomarkers market is significantly influenced by a complex interplay of regulatory, technological, and economic factors. Regulated by a harmonized yet stringent environment that emphasizes clinical evidence and companion diagnostics, the market faces evolving compliance standards such as the In Vitro Diagnostic Regulation (IVDR), which imposes high costs and potential transitional delays for new entrants. Technologically, the integration of next-generation sequencing, liquid biopsies, and artificial intelligence is driving market expansion by enhancing diagnostic accuracy and shortening turnaround times, though these advancements necessitate substantial up-front investments in digital infrastructure and specialized personnel. Economically, while robust government backing through the NHS and initiatives like Genomics England sustain high demand, the market faces significant headwinds from reimbursement challenges, high testing costs, and a critical shortage of skilled pathologists and molecular oncologists. These economic pressures, combined with the high capital investment required for advanced molecular products, can restrain profitability and limit the adoption of cutting-edge equipment in smaller clinical facilities.
Current and Emerging Trends in the United Kingdom Cancer Biomarkers Market
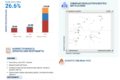
The United Kingdom cancer biomarkers market is undergoing a rapid transformation driven by the integration of artificial intelligence for clinical utility and the widespread adoption of liquid biopsy technologies for non-invasive monitoring. These trends are evolving quickly, as evidenced by the NHS’s 2025 national rollout of circulating tumor DNA tests for lung and breast cancer patients and a projected CAGR of approximately 7.93% to 9.2% through the mid-2030s. Furthermore, the market is shifting toward personalized oncology and the expansion of genetic profiling, which currently accounts for over 68% of demand. While breast cancer remains the largest segment, lung cancer is emerging as the fastest-growing area due to increasing research investments and the implementation of national biomarker-based treatment pathways. Additionally, the move toward value-based oncology and strategic government funding for diagnostic R&D centers are reshaping the industry landscape to meet the needs of a rising cancer burden.
Technological Innovations and Disruption Potential in the United Kingdom Cancer Biomarkers Market
Technological innovations such as next-generation sequencing (NGS), liquid biopsies, and multi-cancer early detection (MCED) tests are gaining significant traction and are poised to disrupt the United Kingdom cancer biomarkers market by enabling non-invasive, precise, and early-stage detection. The integration of artificial intelligence and machine learning is further transforming the industry by streamlining automated laboratory workflows, enhancing the interpretation of complex genomic and proteomic datasets, and assisting in clinical decision support for personalized treatment. Additionally, the development of novel tools like breath biopsies for volatile organic compounds, the iKnife for real-time surgical analysis, and point-of-need testing platforms are decentralizing oncology care and improving patient outcomes through rapid, technology-driven diagnostic insights.
Short-Term vs. Long-Term Trends in the United Kingdom Cancer Biomarkers Market
In the United Kingdom cancer biomarkers market, the initial surge in rapid diagnostic testing demand following the COVID-19 pandemic is viewed as a short-term phenomenon that has stabilized, whereas the integration of precision medicine into routine oncology care represents a long-term structural transformation. The move toward personalized medicine, characterized by the rising adoption of next-generation sequencing and liquid biopsies, is a permanent shift driven by government initiatives through the NHS and a record number of cancer diagnoses requiring targeted therapies. Similarly, the implementation of artificial intelligence for digital pathology and automated result interpretation is a fundamental change aimed at addressing chronic staffing shortages and reducing long turnaround times in laboratories. Other enduring structural trends include the expansion of companion diagnostics and the growing focus on early detection programs for breast and lung cancer, which are sustained by the UK’s long-term commitment to improving cancer survival rates and healthcare innovation.