The United Kingdom autoinjectors market is a rapidly expanding sector of the healthcare industry, valued at over 500 million USD in 2025 and projected to maintain a high compound annual growth rate driven by a significant shift toward home-based care and self-administration of biologic therapies. The landscape is characterized by the increasing prevalence of chronic conditions such as anaphylaxis, diabetes, and rheumatoid arthritis, which necessitate frequent, precise dosing and have led to a high demand for disposable, prefilled devices that enhance patient compliance. Technological advancements are a major hallmark of the market, with ongoing innovations in connected smart injectors for adherence monitoring and the development of large-volume wearable platforms for complex oncology and immunology treatments. While the market is influenced by the National Health Service’s strategic focus on reducing hospital visits and decarbonizing medical device procurement, it remains a competitive environment for global leaders like Pfizer, BD, and Owen Mumford, who are increasingly prioritizing human factors engineering and patient-centric designs to meet stringent regulatory standards and improve user safety.
Key Drivers, Restraints, Opportunities, and Challenges in the United Kingdom Autoinjectors Market
The United Kingdom autoinjectors market is primarily driven by the rising prevalence of chronic and autoimmune diseases such as diabetes, rheumatoid arthritis, and anaphylaxis, alongside a strong healthcare infrastructure that supports a shift toward self-administered, patient-centric drug delivery. Significant growth opportunities exist in the integration of artificial intelligence for personalized dose optimization and the development of sustainable, reusable devices to meet increasing demand for cost-efficient treatments. However, the market faces notable restraints such as high device costs and stringent regulatory requirements that have become more complex following the UK’s exit from the European Union. Key challenges include persistent supply chain vulnerabilities, potential product recalls due to manufacturing defects, and the risk of regulatory divergence leading to delays in the availability of innovative medical devices for UK patients.
Customer Segmentation, Needs, Preferences, and Buying Behavior in the United Kingdom Autoinjectors Market
The target customers for the United Kingdom autoinjectors market primarily include patients managing chronic and acute conditions such as anaphylaxis, rheumatoid arthritis, multiple sclerosis, and diabetes, as well as healthcare providers and the National Health Service (NHS). These customers prioritize convenience, safety, and ease of use, showing a strong preference for disposable, pre-filled devices that facilitate home-based self-administration and reduce the need for hospital visits. Their purchasing behavior is heavily influenced by NHS-supported self-injection programs and clinical recommendations, particularly for life-saving epinephrine devices, while a growing segment of consumers is increasingly utilizing online pharmacies for convenience and recurring refills. Additionally, there is an emerging trend of consumers seeking weight loss medications through both regulated and unregulated digital channels, driven by social media influence and the desire for improved accessibility.
Regulatory, Technological, and Economic Factors Impacting the United Kingdom Autoinjectors Market
The United Kingdom autoinjectors market is significantly influenced by a complex interplay of regulatory, technological, and economic factors that shape entry and growth. From a regulatory perspective, the Medicines and Healthcare Products Regulatory Agency (MHRA) maintains stringent safety and efficacy standards, and the post-Brexit transition to a new UK-specific medical device framework presents both a risk of compliance delays and an opportunity to create a more streamlined, specialist system tailored to National Health Service (NHS) needs. Technologically, the market is being disrupted by the integration of smart technologies, such as connectivity for dose tracking and AI-driven analytics, as well as the development of wearable on-body injectors for high-volume biologics, though these advancements necessitate substantial investments in cybersecurity and data privacy. Economically, while the rising prevalence of chronic conditions and anaphylaxis sustains high demand, profitability is challenged by the high capital investment required for manufacturing and the presence of alternative drug delivery modes like inhalers and oral medications. Furthermore, while the NHS provides a robust data asset for innovation, the market must navigate potential labor shortages and the high costs of advanced device components to ensure long-term competitiveness.
Current and Emerging Trends in the United Kingdom Autoinjectors Market
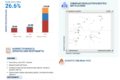
The United Kingdom autoinjectors market is undergoing a rapid evolution driven by the shift toward home-based healthcare and the rising adoption of biologic and biosimilar therapies for chronic conditions. Current trends include a strong preference for disposable, single-use devices and the dominance of the anaphylaxis segment, which is fueled by increasing prescriptions for adrenaline autoinjectors. Emerging trends are characterized by the development of large-volume wearable injectors for complex treatments, the integration of smart, connected technologies for adherence monitoring, and a growing focus on human factors engineering to improve device ergonomics. These trends are evolving quickly, as evidenced by the market’s projected compound annual growth rate (CAGR) of 19.8% through 2033 and the increasing demand for sustainable, low-carbon device alternatives.
Technological Innovations and Disruption Potential in the United Kingdom Autoinjectors Market
Technological innovations such as smart connectivity and digital integration are gaining significant traction and are poised to disrupt the United Kingdom autoinjectors market by enhancing patient adherence through real-time dose tracking and remote monitoring. The adoption of AI-powered platforms and sensors allows these devices to analyze user behavior and send physiological feedback, transforming them into intelligent, patient-centric tools. Additionally, advancements in electromechanical designs and needle-free technologies, such as Crossject’s Zeneo, are improving dose accuracy and safety while reducing needle phobia. The rise of reusable autoinjector platforms, which utilize replaceable drug cartridges and digital add-ons like the FDA-cleared SmartPilot, further disrupts the industry by offering sustainable, cost-effective alternatives to traditional disposable models.
Short-Term vs. Long-Term Trends in the United Kingdom Autoinjectors Market
In the United Kingdom autoinjectors market, temporary spikes in demand for emergency adrenaline devices due to seasonal allergy surges or supply chain resets are viewed as short-term phenomena, whereas several other trends represent long-term structural shifts. The move toward home-based care and self-administration is a permanent transformation driven by the NHS’s strategic focus on reducing hospital visits and the rising prevalence of chronic conditions like diabetes and rheumatoid arthritis. Similarly, the integration of digital connectivity and “smart” sensors into device platforms is a fundamental shift aimed at improving patient adherence and enabling remote clinical oversight. Other enduring structural changes include the rapid expansion of large-volume subcutaneous injectors and the adoption of sustainable, low-carbon device materials, which are fueled by the long-term growth of the biologics pipeline and national healthcare decarbonization targets.